In a neutral atom, the number of electrons equals the number of protons, which means that neutral arsenic atoms have 33 electrons. The sum of the superscripts is equal to the atomic number. Its atomic number is 33, which is the number of protons in the nuclei of its atoms. The electron configuration for every element above Ar has the 1s 2 2s 2 2p 6 orbital configuration. Ito ay isang koleksyon ng mga diagram ng mga atomo na nagpapakita ng mga bilang ng mga proton, neutron, at mga electron na nasa atom o isotope ng isang elemento. Click on 'Element Atomic Number', 'Element Symbol', 'Element Name' and 'Element Electron Configuration ' headers to sort. The electron configuration of an element with an atomic number greater than 18 cannot be properly determined according to the Bohr atomic model.
#AS ELEMENT ELECTRON CONFIGURATION FULL#
For the undiscovered eighth-row elements, mixing of configurations is expected to be very important, and sometimes the result can no longer be well-described by a single configuration. Explanation: As is the chemical symbol for the element arsenic. This Electron Configuration table gives the Electron Configuration of all the elements of periodic table. The oganesson full electron configuration will be 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 4f14 5s2 5p6 5d10 5f14 6s2 6p6 6d10 7s2 7p6. In many cases, multiple configurations are within a small range of energies and the irregularities shown below do not necessarily have a clear relation to chemical behaviour. Note that these electron configurations are given for neutral atoms in the gas phase, which are not the same as the electron configurations for the same atoms in chemical environments. However there are numerous exceptions for example the lightest exception is chromium, which would be predicted to have the configuration 1s 2 2s 2 2p 6 3s 2 3p 6 3d 4 4s 2, written as 3d 4 4s 2, but whose actual configuration given in the table below is 3d 5 4s 1. Electron configurations of elements beyond hassium (element 108) have never been measured predictions are used below.Īs an approximate rule, electron configurations are given by the Aufbau principle and the Madelung rule. For each atom the subshells are given first in concise form, then with all subshells written out, followed by the number of electrons per shell. It lists the orbital symbols sequentially. The electronic configuration can be written as 1s 2 2s 2 2p 6 3s 2 3p 6 4s 1.
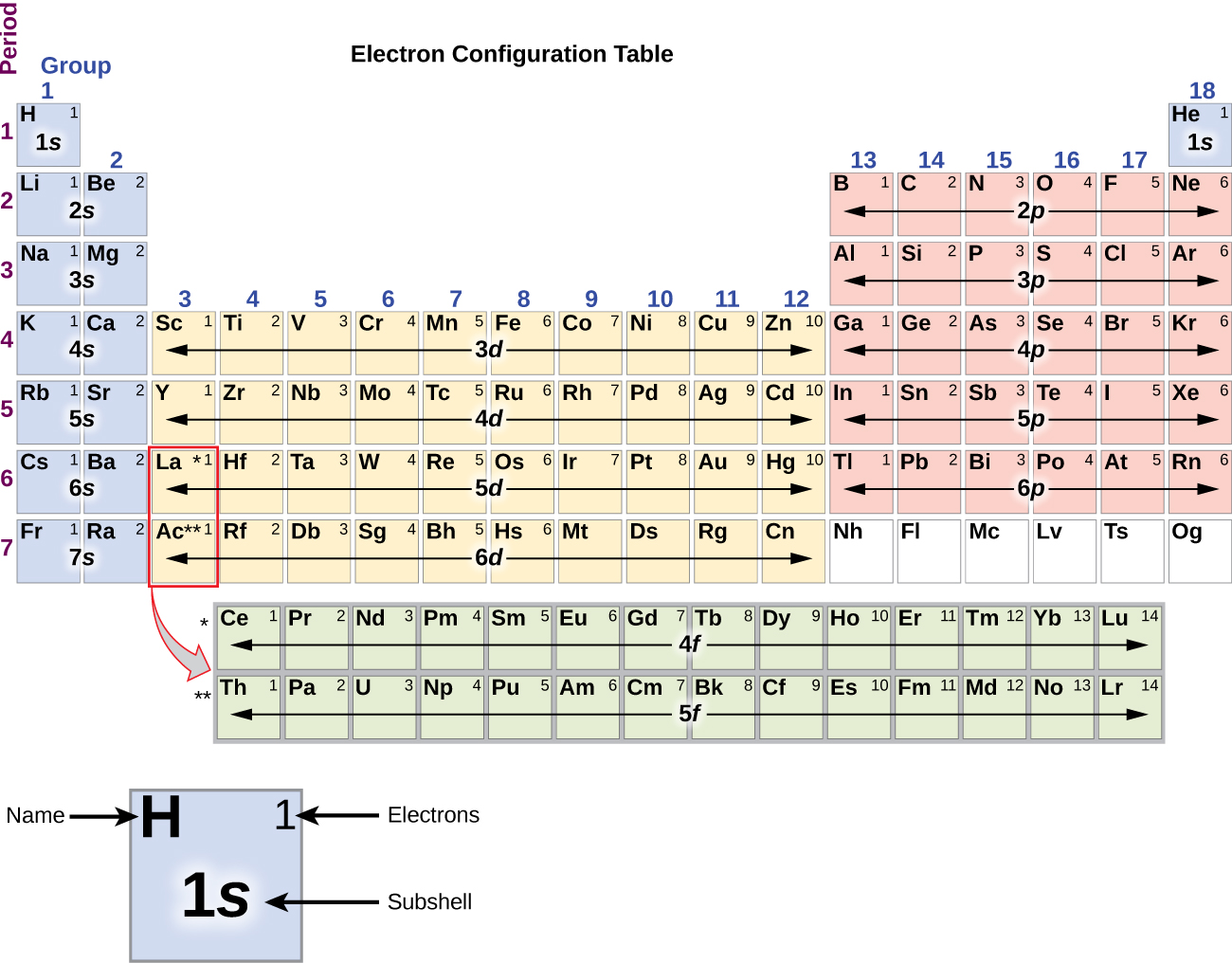
And has 19 electrons which will be placed in s and p sub-shell.
#AS ELEMENT ELECTRON CONFIGURATION HOW TO#
This video might also be useful, it describes how to write short-cut electron configurations. The potassium element has atomic number 19. Once you practice a few different examples writing electron configurations will become pretty easy. This page shows the electron configurations of the neutral gaseous atoms in their ground states. As previously mentioned, electron configuration is a particular distribution of electrons among available orbitals. This video explains how to use the Aufbau principle and a diagonal diagram to write electron configurations.
